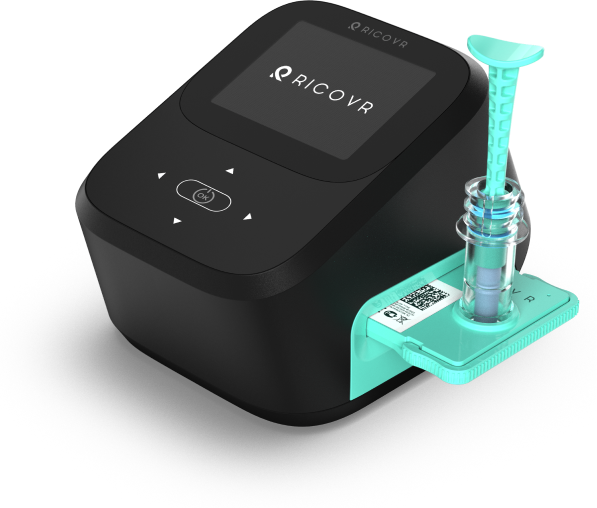
XALIVA® THC
Key Benefits

Rapid Results
Screen quickly in minutes not days
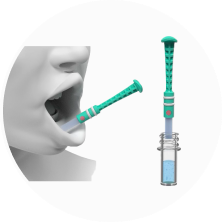
Non-Invasive
Saliva collection uses direct observation while maintaining privacy and fairness

High Accuracy
Detects all forms of ingested THC (e.g. smoking, vaping, edibles, etc)
Advantages of Xaliva® THC in the Workplace
Industries
Construction
Manufacturing
Warehousing
Logistics
Transportation
Food & Beverage
Oil & Gas
Scenarios
Random
Reasonable
Suspicion
Post-Accident
Periodic/
Regular Testing
Return-to-Duty
Testing
Follow-Up Testing
Benefits
Safety
Productivity
Efficiency
Quality
Compliance & Risk
Retention
Employee Satisfaction
We Serve
Our Xaliva® THC device solution can be used by many stakeholders across various industries to ensure workplace safety

Employers
HR professionals and supervisors can easily conduct on-site "in-house" testing for various scenarios according to their HR Policy.
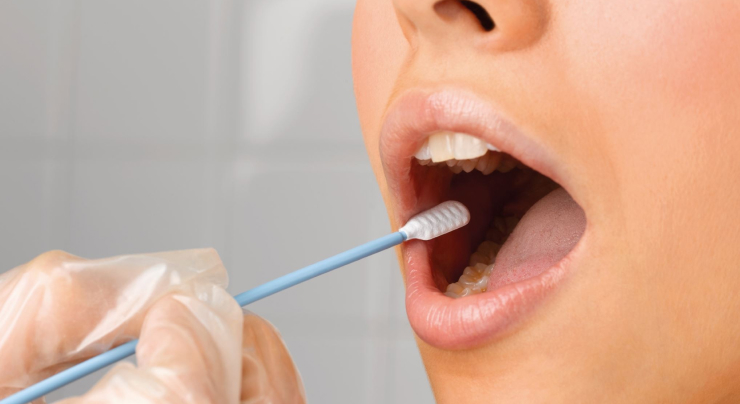
Third Party Administrators (TPAs)
TPAs and mobile specimen collectors can easily schedule on-site saliva sample collections and run tests providing results in minutes.